A team of HLI staff and researchers (Drs. Ma’en Obeidat – Assistant Professor, Anthony Tam – Postdoctoral Fellow, and Tony Yang – Research & Industry Partnerships Manager) were recently invited to tour Chinook Therapeutics, a newly established local pharmaceutical discovery company.
Launched from San Diego-headquartered, parent drug discovery engine Inception Sciences, Chinook Therapeutics is a biotechnology company developing precision medicines for kidney diseases. Since 2011, Inception Sciences has research sites in Vancouver, Montreal, and San Diego, with notable successes with pharma acquisitions.
Welcomed by Joyce Wu – Senior Research Associate, Dr. Jennifer Cox – Assistant Director, and Dr. Renata Oballa – Vice President of Medicinal Chemistry, our HLI team had the unique opportunity to tour the Chinook Therapeutics research facilities and discuss potential educational opportunities for trainees of the Career Paths for Researchers (CPR) program. The company’s initiatives, which include multi-disciplinary and integrated medicinal chemistry, biology, bioinformatics, drug metabolism and pharmacokinetics, and target validation studies, were shared and potential collaboration opportunities were discussed.
Thank you Chinook Therapeutics for hosing us and we look forward to developing the next CPR-Chinook trainee event. Stay tuned!
HLI Director Dr. Don Sin and a team of HLI researchers, including 5 trainees, were recently invited to attend a tour of IKOMED Technologies Inc. (www.ikomed.com, founded in 2011), an event organized by the HLI Career Paths for Researchers (CPR) program.
The researchers learned more about the company’s commercialization activities and discussed ongoing research collaborations. One of the IKOMED team’s current projects is the commercialization of their innovative technology designed to reduce harmful exposures to X-ray radiation, for patients and staff, during surgical procedures involving the use of fluoroscopy (a type of high speed X-ray imaging that creates a X-ray “movie”).
Our hosts – CEO Eran Elizur, and Director of Program Management Kim Wolff – showcased their FDA-approved AI-enabled radiation reduction technology which was recently cleared by the FDA and has begun commercial sales. The team also discussed the next steps of an ongoing research collaboration that aims to leverage novel radiofrequency technology for the treatment of patients with emphysema, a debilitating and cureless chronic lung disease. Thank you and congratulations to the IKOMED team!
Airway remodeling, which refers to changes to the structure of the airways, occurs in asthma and is linked to reduced lung function and a greater use of asthma medications. Collagen, in particular, is an important component of the airway structure that accumulates more in asthmatic airways compared to the airways of healthy individuals.

Using state-of-the-art microscopy techniques, Dr. Tillie Hackett and her research team at the Centre for Heart Lung Innovation set out to image the collagen and elastin fiber s that make up the air way in order to better understand airway remodeling. What the team discovered was that collagen fibers are more disorganized and fragmented in the airway walls of asthmatic individuals compared to individuals without asthma. This not only provides a novel drug target for asthma treatment, but also supports the use of these advanced imaging tools to monitor airway remodeling throughout disease progression and during treatment. These results were recently published in the American Journal of Respiratory and Critical Care Medicine and featured on Extracellular Matrix News.
This research was made possible by the purchase of a new non-linear optimal microscope, the first super-resolution microscope of its kind in Canada, through funds from the Canadian Foundation of Innovation and St. Paul’s Hospital Foundation.
The April issue of the Lobe and Mail, HLI’s newsletter, is now available! Please click the PDF link below to learn about exciting research findings from our Centre, meet some of our HLI personnel, and find out what we have been up to in the city and across the Atlantic Ocean!
Stay connected with the HLI and subscribe to our newsletter by emailing communications@hli.ubc.ca.
The first week of a newborn’s life is a time of the most rapid biological change as the baby adapts to living outside the womb, suddenly exposed to new challenges such as bacteria and viruses. Yet surprisingly little is known about these early changes. An international research study, co-led by University of British Columbia and Boston Children’s Hospital, has pioneered a technique to get huge amounts of data from a tiny amount of newborn blood, allowing for the most comprehensive data analysis to date.
“We found thousands of changes over the first week of life, including changes in gene expression and parts of the immune defense pathways.”
Casey Shannon, co-lead author and a computational biologist at the Prevention of Organ Failure (PROOF) Centre of Excellence at St. Paul’s Hospital.
Published today in Nature Communications, this study revealed key molecular changes in the first week of newborns’ life, establishing a common developmental pathway to further our understanding of newborn health and the impact of vaccines. This study showcases important contributions from researchers at the Centre for Heart Lung Innovation (HLI) and PROOF Centre, with Casey Shannon as a co-lead author, Daniel He and Dr. Amrit Singh as co-authors, and Dr. Scott Tebbutt as a senior author. One of the key computational approaches that was used for this study was developed by Dr. Singh during his Ph.D.

The researchers piloted their technique with a group of newborns from The Gambia in West Africa after first obtaining permission from village elders and informed consent from mothers in local languages. Importantly, they then validated the approach with a second group of newborns from Papua New Guinea (Australasia). What they found was that the two independent groups of babies shared a common, highly dynamic developmental trajectory—suggesting that the molecular changes do not occur at random, but instead follow an age-specific pathway which may be used to monitor the impact of life-saving interventions.
Chronic obstructive pulmonary disease (COPD) affects over 2 million Canadians and is the major cause of hospital admissions of adult males and increasingly females over 40 years of age in Canada.
More than 50 years ago, Canadians working at McGill University in Montreal were the first to show that the major site of airflow obstruction in COPD is located in the small airways in the lung. Last year, researchers at the Centre for Heart Lung Innovation, located at St Paul’s Hospital, have shown using the latest imaging technologies with unprecedented resolution that the small airways deep within the lungs are destroyed before any clinical symptoms or abnormalities can be observed (Lancet Respiratory Medicine).
We know now that once airways are damaged, it is generally irreversible and will continue to progress, even for those who quit tobacco products. Unfortunately, today’s available treatments for COPD are mostly directed at relieving symptoms by dilating constricted airways or treating inflammation, but nothing is designed to reverse the disease responsible for the airway obstructions. Therefore, early detection of airway abnormalities would be key for any successful treatment.
Last week, researchers at the HLI in collaboration with clinicians at University of Michigan and Temple University, have published a new study which for the first time validated a novel imaging biomarker that detects small airways disease using conventional CT imaging. Dr. Dragoş Vasilescu, a Research Associate at the HLI and an expert in ultra-high resolution micro CT imaging, demonstrated by using lung specimen from patients who received a lung transplant that there was indeed a strong agreement between the clinically available imaging biomarker and the precise high resolution measurements. For his work, Dr. Vasilescu used a micro CT scanner located at the HLI and software that he and his colleagues have developed to image and measure the disease present in the smallest airways in the lung. This new study has been published in the American Journal of Respiratory and Critical Care Medicine.
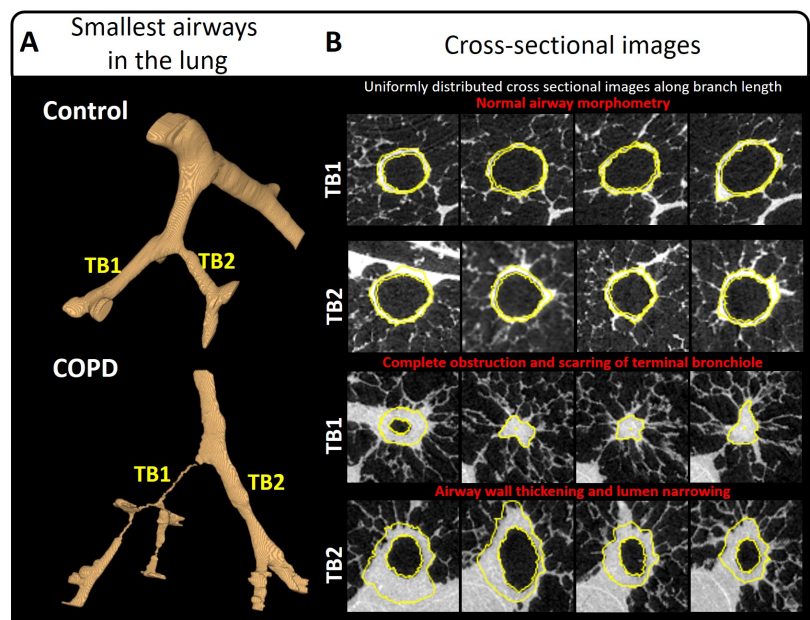
It is the hope of the whole group that this novel technique provides a way to accurately identify patients who are most likely to progress to symptomatic lung disease before they can be diagnosed with traditional methods such as pulmonary function tests, ensuring earlier and more effective treatment for the patients. This study could revolutionize how new therapies for COPD and potentially other lung diseases are developed.
Read more about it in the UBC Pathology Newsletter and in the Daily Scan.
Podocalyxin is a protein that is normally expressed in blood vessels, and is particularly highly expressed in the blood vessels of the brain. In this study published in the Proceedings of the National Academcy of Sciences, HLI researcher Dr. Kelly McNagny and his team discovered the important role of podocalyxin in maintaining the integrity of the blood brain barrier during acute inflammation.
These results are significant as the blood brain barrier is tightly regulated, separating the brain from circulating blood. A healthy blood brain barrier is essential for maintaining normal brain function. Disruption of this barrier can promote the damage of brain tissue during infections and neurological
diseases, such as stroke, epilepsy, and multiple sclerosis. In addition, understanding how the blood brain barrier is regulated will not only provide important insights into these neurological diseases, but may also allow researchers to effectively deliver therapeutics to the brain.
Read the full paper here.
Aso featured in UBC News.
The first issue of our newly redesigned HLI newsletter, The Lobe and Mail, is out! This issue features our new Director, exciting research updates, as well as community and research events and much more.
