Alzheimer’s disease is a progressive, degenerative brain disease that causes memory impairment and deterioration of thinking ability. Current diagnosis depends on physician evaluation of the signs and symptoms of neurodegeneration, coupled with traditional imaging. By the time most people are diagnosed, they often already have significant mental decline and cognitive impairment. Accurate and timely diagnosis is important to ensure that a patient receives the right treatment (which is most effective if provided early), and that, together with family and caregivers, they have the ability to plan for the future.
Measuring the levels of two proteins (amyloid-beta and tau) in cerebrospinal fluid has been shown to help correctly identify those with Alzheimer’s disease. Such testing, which can be performed early-on in the disease course, can predict whether mild symptoms are likely to progress to dementia. Although in
many countries, measuring amyloid-beta and tau is now part of routine practice, in Canada, there still exist barriers that prevent implementation of this test in routine care.
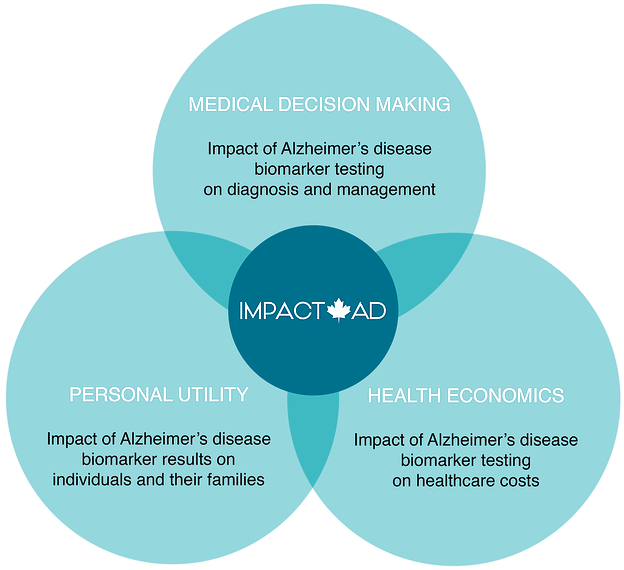
HLI Principal Investigator Dr. Mari DeMarco is the lead investigator on IMPACT-AD, a Canada-wide study that aims to bridge the gap between diagnostic accuracy studies and clinical utilization and implementation of these biomarker tests. IMPACT-AD will determine how the testing for these Alzheimer’s disease biomarkers impacts medical and personal decision making, as well as health care costs. The goal of the study is to inform positive change in the health care system and improve care and support for patients living with Alzheimer’s, and their families.
If results are positive, this could pave the way for biomarker testing to
become part of routine Alzheimer’s disease care in Canada. Learn more
about the study in The Daily Scan: here and here. For more information,
visit: https://www.impactad.org/
the Michael Smith Foundation for Health Research, the UBC Djavad Mowafaghian Centre for Brain Health and the Faculty of Medicine, the Women’s Brain Health Initiative, and the St. Paul’s Foundation.
